Unveil The Secrets Of Electronegativity Order: Unlocking Chemical Mysteries
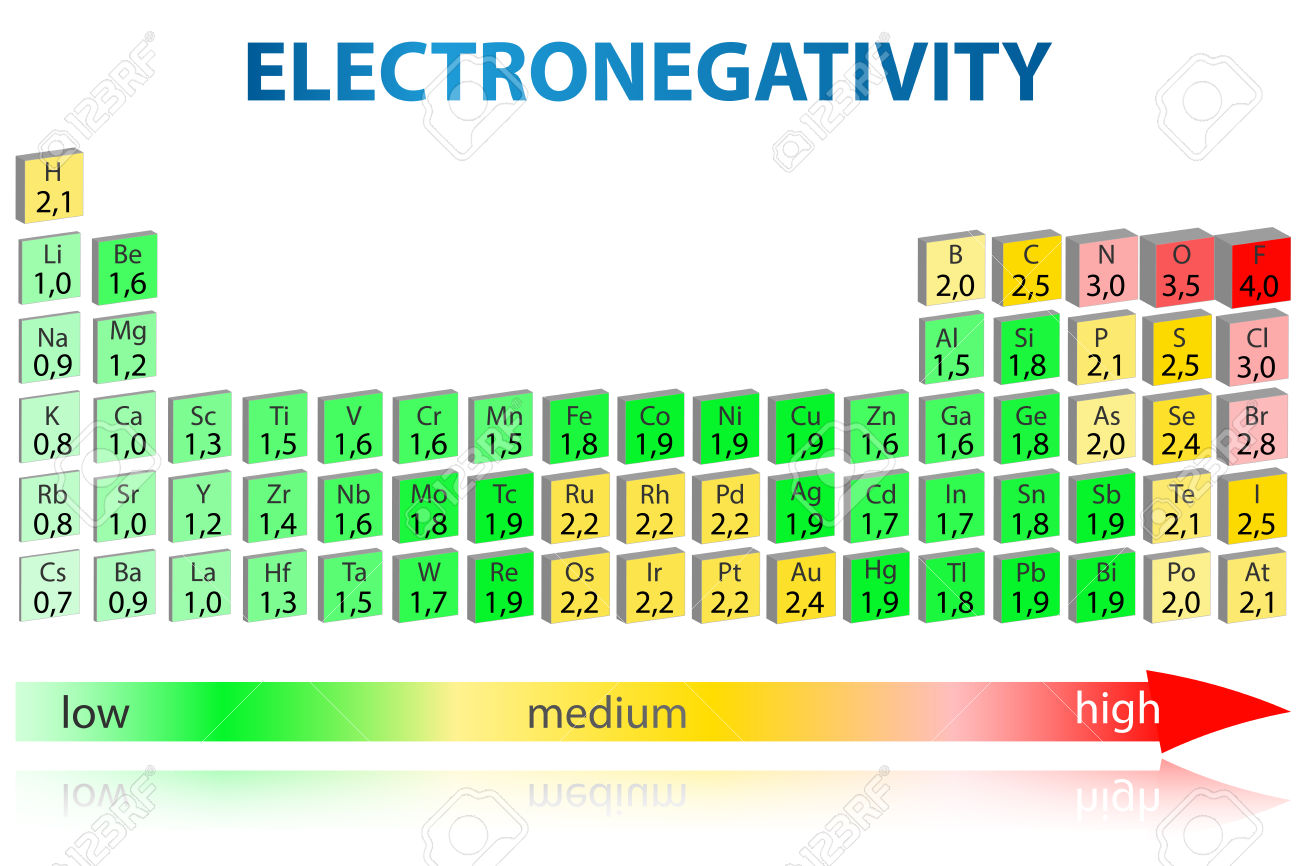
Electronegativity is a chemical property that describes the ability of an atom to attract electrons towards itself. Electronegativity order is the ranking of elements based on their electronegativity values. The most electronegative element is fluorine, followed by oxygen, nitrogen, chlorine, and bromine.
Electronegativity order is important because it can be used to predict the chemical properties of elements. For example, elements with high electronegativity values are more likely to form ionic bonds, while elements with low electronegativity values are more likely to form covalent bonds. Electronegativity order can also be used to explain the periodic trends in electronegativity.
The concept of electronegativity was first proposed by Linus Pauling in 1932. Pauling's electronegativity scale is still the most widely used electronegativity scale today. Electronegativity order is a valuable tool for chemists, and it is used in a variety of applications, including predicting the chemical properties of elements, explaining periodic trends, and designing new materials.
Electronegativity Order
Electronegativity order is a fundamental concept in chemistry that describes the relative ability of atoms to attract electrons. It is a key factor in determining the chemical properties of elements and compounds.
- Periodic Trend: Electronegativity generally increases from left to right across a period and decreases from top to bottom within a group.
- Bond Polarity: The difference in electronegativity between two atoms determines the polarity of a chemical bond.
- Ionic vs. Covalent: Highly electronegative elements tend to form ionic bonds, while less electronegative elements tend to form covalent bonds.
- Molecular Shape: Electronegativity influences the shape of molecules by affecting the distribution of electrons.
- Reactivity: Electronegative elements are more reactive and readily participate in chemical reactions.
- Electronegativity and Size: Smaller atoms tend to be more electronegative than larger atoms.
- Electronegativity and Oxidation State: Oxidation state can affect the electronegativity of an atom.
- Electronegativity and Ionization Energy: Electronegativity is related to ionization energy, with more electronegative elements having higher ionization energies.
- Electronegativity and Electron Affinity: Electronegativity is also related to electron affinity, with more electronegative elements having higher electron affinities.
- Applications: Electronegativity order is used in various fields, including predicting chemical reactivity, designing new materials, and understanding biological processes.
In summary, electronegativity order is a crucial concept that helps us understand and predict the chemical behavior of elements and compounds. It is a fundamental property that influences a wide range of chemical phenomena, from bond formation to molecular structure and reactivity.
Periodic Trend
The periodic trend of electronegativity is closely related to the concept of electronegativity order. Electronegativity order refers to the ranking of elements based on their electronegativity values, with fluorine being the most electronegative element and francium being the least electronegative element.
The periodic trend of electronegativity can be explained by considering the atomic structure of elements. As we move from left to right across a period, the number of protons in the nucleus increases, which increases the attraction for electrons. This results in a higher electronegativity. On the other hand, as we move down a group, the number of electron shells increases, which increases the distance between the nucleus and the valence electrons. This results in a lower electronegativity.
The periodic trend of electronegativity has important implications in chemistry. For example, it helps us understand the formation of chemical bonds. Elements with high electronegativity values tend to form ionic bonds, while elements with low electronegativity values tend to form covalent bonds. Additionally, the electronegativity of an element can influence the polarity of a molecule, its reactivity, and its other chemical properties.
In summary, the periodic trend of electronegativity is a fundamental concept in chemistry that helps us understand and predict the chemical behavior of elements and compounds. It is closely related to the concept of electronegativity order, which provides a quantitative measure of the electronegativity of different elements.
Bond Polarity
Electronegativity order plays a crucial role in determining bond polarity. Bond polarity refers to the uneven distribution of electrons in a chemical bond, resulting in a partial positive charge on one atom and a partial negative charge on the other atom. The greater the difference in electronegativity between two atoms, the more polar the bond will be.
For example, in a bond between hydrogen and chlorine, chlorine is more electronegative than hydrogen. This means that chlorine attracts the shared electrons more strongly, resulting in a polar bond with a partial negative charge on the chlorine atom and a partial positive charge on the hydrogen atom.
Bond polarity is important because it influences many chemical properties, such as solubility, reactivity, and molecular shape. Polar molecules tend to be more soluble in polar solvents and can participate in hydrogen bonding. Bond polarity also affects the reactivity of molecules, as polar bonds can interact with other polar molecules or ions.
In summary, electronegativity order is closely connected to bond polarity. The difference in electronegativity between two atoms determines the polarity of the bond they form. Bond polarity, in turn, has a significant impact on the chemical properties of molecules.
Ionic vs. Covalent
Electronegativity order plays a crucial role in determining the type of chemical bond that will form between two atoms. Ionic bonds are formed when there is a large difference in electronegativity between two atoms, while covalent bonds are formed when the difference in electronegativity is small.
Ionic bonds are typically formed between a metal and a nonmetal. The metal atom loses one or more electrons to the nonmetal atom, resulting in the formation of positively charged metal ions and negatively charged nonmetal ions. The electrostatic attraction between the oppositely charged ions holds the ionic compound together.
Covalent bonds are typically formed between two nonmetal atoms. The atoms share one or more pairs of electrons, resulting in the formation of a covalent bond. The strength of the covalent bond depends on the number of shared electrons.
The distinction between ionic and covalent bonds is important because it affects the properties of the compound. Ionic compounds are typically hard and brittle, while covalent compounds are typically soft and flexible. Ionic compounds are also good conductors of electricity, while covalent compounds are typically poor conductors of electricity.
Electronegativity order can be used to predict the type of bond that will form between two atoms. For example, sodium is a highly electropositive element, while chlorine is a highly electronegative element. This means that sodium will readily lose an electron to chlorine, resulting in the formation of an ionic bond.
In summary, electronegativity order is a key factor in determining the type of chemical bond that will form between two atoms. Ionic bonds are formed when there is a large difference in electronegativity, while covalent bonds are formed when the difference in electronegativity is small.
Molecular Shape
The electronegativity of an atom affects the distribution of electrons within a molecule, which in turn influences the molecular shape. Electronegative atoms attract electrons toward them, creating a more localized electron distribution. This can lead to the formation of polar covalent bonds, in which the electrons are not shared equally between the atoms. The polarity of the bonds can then affect the overall shape of the molecule.
For example, consider the molecule of water (H2O). Oxygen is more electronegative than hydrogen, so the electrons in the O-H bonds are pulled toward the oxygen atom. This creates a polar covalent bond, with a partial negative charge on the oxygen atom and a partial positive charge on the hydrogen atoms. The polarity of the O-H bonds causes the water molecule to have a bent shape, with the hydrogen atoms positioned at an angle to each other.
The shape of molecules is important because it can affect their physical and chemical properties. For example, the bent shape of the water molecule allows it to form hydrogen bonds with other water molecules, which is responsible for the unique properties of water, such as its high surface tension and high specific heat capacity.
Electronegativity order is a useful tool for predicting the shape of molecules. By understanding the electronegativity of the atoms involved, we can predict the polarity of the bonds and the overall shape of the molecule.
Reactivity
Electronegative elements are more reactive because they have a strong tendency to attract electrons. This means that they are more likely to participate in chemical reactions in which they can gain electrons. For example, fluorine is the most electronegative element and is highly reactive. It readily reacts with other elements to form compounds, such as hydrogen fluoride (HF) and sodium fluoride (NaF).
Electronegativity order is important in predicting the reactivity of elements. The more electronegative an element is, the more reactive it is. This is because electronegative elements are more likely to gain electrons and form chemical bonds. For example, fluorine is more reactive than chlorine, which is more reactive than bromine, and so on.
The reactivity of electronegative elements has many practical applications. For example, fluorine is used in the production of uranium hexafluoride (UF6), which is used in the enrichment of uranium for nuclear power and nuclear weapons. Chlorine is used in the production of polyvinyl chloride (PVC), which is a widely used plastic. Bromine is used in the production of silver bromide (AgBr), which is used in photographic film.
In summary, electronegative elements are more reactive and readily participate in chemical reactions. Electronegativity order is important in predicting the reactivity of elements. The reactivity of electronegative elements has many practical applications.
Electronegativity and Size
The electronegativity of an atom is affected by its size. Smaller atoms tend to be more electronegative than larger atoms. This is because smaller atoms have a higher nuclear charge density, which means that the electrons are held more tightly to the nucleus. As a result, smaller atoms have a stronger attraction for electrons and are more likely to attract electrons from other atoms.
The connection between electronegativity and size is important because it helps us to understand the chemical properties of elements. For example, the high electronegativity of fluorine is due to its small size. Fluorine is the most electronegative element and is highly reactive. It readily reacts with other elements to form compounds, such as hydrogen fluoride (HF) and sodium fluoride (NaF).
In contrast, the low electronegativity of cesium is due to its large size. Cesium is the least electronegative element and is very unreactive. It does not readily react with other elements and is often used in electronic devices because of its low reactivity.
The relationship between electronegativity and size is a fundamental concept in chemistry. It helps us to understand the chemical properties of elements and to predict how they will react with other elements.
Electronegativity and Oxidation State
The oxidation state of an atom is the hypothetical charge that the atom would have if all of its bonds were ionic. Oxidation state can affect the electronegativity of an atom because it changes the distribution of electrons around the atom. Atoms with higher oxidation states have a greater attraction for electrons, and are therefore more electronegative.
- Effect of Oxidation State on Electronegativity
The oxidation state of an atom can have a significant effect on its electronegativity. For example, the electronegativity of chlorine increases from 2.5 to 3.0 when it goes from an oxidation state of -1 to an oxidation state of +1.
- Electronegativity and Chemical Bonding
The electronegativity of an atom can affect the type of chemical bonds that it forms. For example, atoms with high electronegativity values tend to form ionic bonds, while atoms with low electronegativity values tend to form covalent bonds.
- Electronegativity and Periodic Trends
The electronegativity of an atom is affected by its position in the periodic table. In general, electronegativity increases from left to right across a period and decreases from top to bottom within a group.
- Applications of Electronegativity
Electronegativity is a useful concept that can be used to understand and predict a wide range of chemical phenomena. For example, electronegativity can be used to predict the polarity of bonds, the solubility of compounds, and the reactivity of elements.
In summary, the oxidation state of an atom can affect its electronegativity. Electronegativity is a fundamental property of atoms that can be used to understand and predict a wide range of chemical phenomena.
Electronegativity and Ionization Energy
Electronegativity and ionization energy are two closely related concepts in chemistry. Electronegativity measures the ability of an atom to attract electrons, while ionization energy measures the energy required to remove an electron from an atom. In general, more electronegative elements have higher ionization energies.
- Ionization Energy and Atomic Size
One of the factors that affects ionization energy is atomic size. Smaller atoms have higher ionization energies than larger atoms. This is because the electrons in smaller atoms are held more tightly to the nucleus.
- Ionization Energy and Nuclear Charge
Another factor that affects ionization energy is nuclear charge. Atoms with a higher nuclear charge have higher ionization energies. This is because the increased nuclear charge attracts the electrons more strongly.
- Electronegativity and Ionization Energy Trends
The trends in ionization energy are similar to the trends in electronegativity. Ionization energy increases from left to right across a period and decreases from top to bottom within a group.
- Applications of Ionization Energy
Ionization energy is a useful concept that can be used to understand and predict a wide range of chemical phenomena. For example, ionization energy can be used to predict the reactivity of elements, the stability of compounds, and the properties of materials.
In summary, electronegativity and ionization energy are two closely related concepts that can be used to understand and predict a wide range of chemical phenomena. The connection between electronegativity and ionization energy is important because it provides a deeper understanding of the electronic structure of atoms and their chemical behavior.
Electronegativity and Electron Affinity
The connection between electronegativity and electron affinity is important because it provides a deeper understanding of the electronic structure of atoms and their chemical behavior. Electronegativity measures the ability of an atom to attract electrons, while electron affinity measures the energy change when an atom gains an electron. More electronegative elements have higher electron affinities because they have a stronger attraction for electrons. This relationship is important for understanding a wide range of chemical phenomena, including bond formation, molecular structure, and chemical reactivity.
For example, the high electronegativity and electron affinity of fluorine make it a highly reactive element. Fluorine readily reacts with other elements to form compounds, such as hydrogen fluoride (HF) and sodium fluoride (NaF). In contrast, the low electronegativity and electron affinity of cesium make it a very unreactive element. Cesium does not readily react with other elements and is often used in electronic devices because of its low reactivity.
The relationship between electronegativity and electron affinity is a fundamental concept in chemistry. It helps us to understand the chemical properties of elements and to predict how they will react with other elements. This understanding is essential for the development of new materials and technologies.
Applications
Electronegativity order is a powerful tool that has a wide range of applications in various fields, including chemistry, materials science, and biology. By understanding the electronegativity of different elements, scientists and engineers can predict chemical reactivity, design new materials with specific properties, and understand biological processes at the molecular level.
One important application of electronegativity order is in predicting chemical reactivity. Electronegativity can be used to predict the type of chemical bonds that will form between two atoms. For example, elements with high electronegativity values tend to form ionic bonds, while elements with low electronegativity values tend to form covalent bonds. This understanding of chemical bonding is essential for predicting the reactivity of different compounds and for designing new materials with specific properties.
Another important application of electronegativity order is in the design of new materials. By understanding the electronegativity of different elements, scientists and engineers can design materials with specific properties, such as high strength, low weight, or high electrical conductivity. For example, electronegativity order has been used to design new alloys with improved mechanical properties and to develop new semiconductor materials for use in electronic devices.
Electronegativity order is also used in the understanding of biological processes at the molecular level. Electronegativity can be used to predict the polarity of molecules, which in turn can affect their solubility, reactivity, and biological activity. For example, the polarity of water molecules is due to the difference in electronegativity between oxygen and hydrogen. This polarity is essential for the many important roles that water plays in biological systems.
In summary, electronegativity order is a powerful tool that has a wide range of applications in various fields, including chemistry, materials science, and biology. By understanding the electronegativity of different elements, scientists and engineers can predict chemical reactivity, design new materials with specific properties, and understand biological processes at the molecular level.
FAQs on Electronegativity Order
Electronegativity order is a fundamental concept in chemistry that describes the relative ability of atoms to attract electrons. It is widely used to predict chemical reactivity, design new materials, and understand biological processes at the molecular level. Here are some frequently asked questions about electronegativity order:
Question 1: What is electronegativity order and how is it determined?
Answer: Electronegativity order is a ranking of elements based on their electronegativity values. Electronegativity is a measure of the ability of an atom to attract electrons. It is determined by considering the atomic number, atomic radius, and shielding effect of an element.
Question 2: How does electronegativity order affect chemical bonding?
Answer: Electronegativity order plays a crucial role in determining the type of chemical bond that will form between two atoms. Elements with large differences in electronegativity tend to form ionic bonds, while elements with small differences in electronegativity tend to form covalent bonds.
Question 3: What are some applications of electronegativity order in materials science?
Answer: Electronegativity order is used in materials science to design new materials with specific properties, such as high strength, low weight, or high electrical conductivity. By understanding the electronegativity of different elements, scientists and engineers can create materials with tailored properties for various applications.
Question 4: How is electronegativity order used in understanding biological processes?
Answer: Electronegativity order is used in biology to understand the polarity of molecules, which in turn affects their solubility, reactivity, and biological activity. For example, the polarity of water molecules is due to the difference in electronegativity between oxygen and hydrogen. This polarity is essential for water's role as a solvent and its involvement in various biological processes.
Question 5: What are some limitations of electronegativity order?
Answer: While electronegativity order is a useful concept, it has some limitations. It does not always accurately predict bond polarity or the type of chemical bond that will form between two atoms. Additionally, electronegativity values can vary depending on the chemical environment and the oxidation state of the atoms involved.
Question 6: How can I learn more about electronegativity order?
Answer: There are numerous resources available to learn more about electronegativity order. You can find detailed explanations in chemistry textbooks, online articles, and scientific papers. Additionally, there are interactive simulations and videos that can help you visualize and understand the concept more clearly.
In summary, electronegativity order is a valuable tool for understanding and predicting chemical behavior. It has wide applications in chemistry, materials science, and biology. While there are some limitations to its use, electronegativity order remains a fundamental concept that helps us unravel the electronic structure and reactivity of elements and compounds.
Transition to the next article section:
Now that we have explored electronegativity order, let's move on to another important chemical concept: bond polarity.
Tips on Understanding Electronegativity Order
Electronegativity order is a fundamental concept in chemistry that describes the relative ability of atoms to attract electrons. Understanding electronegativity order is crucial for predicting chemical behavior, designing new materials, and comprehending biological processes. Here are some tips to help you grasp and apply the concept of electronegativity order:
Tip 1: Relate Electronegativity to Periodic Trends
Electronegativity generally increases from left to right across a period and decreases from top to bottom within a group in the periodic table. This trend can help you quickly estimate the electronegativity of an element based on its position in the table.
Tip 2: Consider Atomic Size and Nuclear Charge
Electronegativity is influenced by both atomic size and nuclear charge. Smaller atoms with a higher nuclear charge tend to be more electronegative. This is because the smaller size allows for a stronger attraction between the nucleus and the electrons.
Tip 3: Visualize Electronegativity Differences
To understand the impact of electronegativity differences, visualize the electron distribution in a bond between two atoms. A large electronegativity difference leads to a more polar bond, where electrons are drawn towards the more electronegative atom. Conversely, a small electronegativity difference results in a less polar or nonpolar bond.
Tip 4: Predict Ionic or Covalent Bonding
Electronegativity order can help you predict the type of chemical bond that will form between two atoms. A large electronegativity difference often leads to ionic bonding, where one atom completely transfers electrons to the other. A small electronegativity difference typically results in covalent bonding, where electrons are shared between the atoms.
Tip 5: Understand Electronegativity and Reactivity
More electronegative elements are generally more reactive. This is because they have a stronger tendency to attract electrons and participate in chemical reactions to achieve a more stable electron configuration.
Tip 6: Apply Electronegativity in Material Design
Electronegativity order is used in materials science to design materials with specific properties. For example, materials with alternating layers of highly electronegative and less electronegative elements can exhibit interesting electronic and optical properties.
Summary
By following these tips, you can develop a deeper understanding of electronegativity order and its applications in chemistry and other fields. Remember to consider periodic trends, atomic size, nuclear charge, and the impact of electronegativity on bond polarity and chemical reactivity. With a solid grasp of electronegativity order, you can unlock a wealth of insights into the behavior of elements and compounds.
Conclusion
Throughout this article, we have explored the concept of electronegativity order, its significance, and its wide-ranging applications in chemistry and beyond. Electronegativity order provides a fundamental understanding of the relative ability of atoms to attract electrons, enabling us to predict chemical behavior, design new materials, and unravel biological processes at the molecular level.
Electronegativity order is a powerful tool that has shaped our understanding of chemical bonding, molecular polarity, and chemical reactivity. It has played a crucial role in the development of new materials with tailored properties, revolutionizing industries and advancing scientific research. By comprehending electronegativity order, we gain insights into the electronic structure and behavior of matter, opening up new avenues for innovation and discovery.